A pulmonary embolism (PE) stops being an abstract clinical threat the moment it impacts someone you love. It turns an ordinary day into a race against the clock — a harrowing experience and condition that claims an American life roughly every six minutes.
Pulmonary embolism is the third most common cause of death from cardiovascular disease after heart attack and stroke, responsible for an estimated 60-100K deaths annually. Despite the scale and severity, treatment has historically relied on approaches that are either passive or risky with significant limitations.
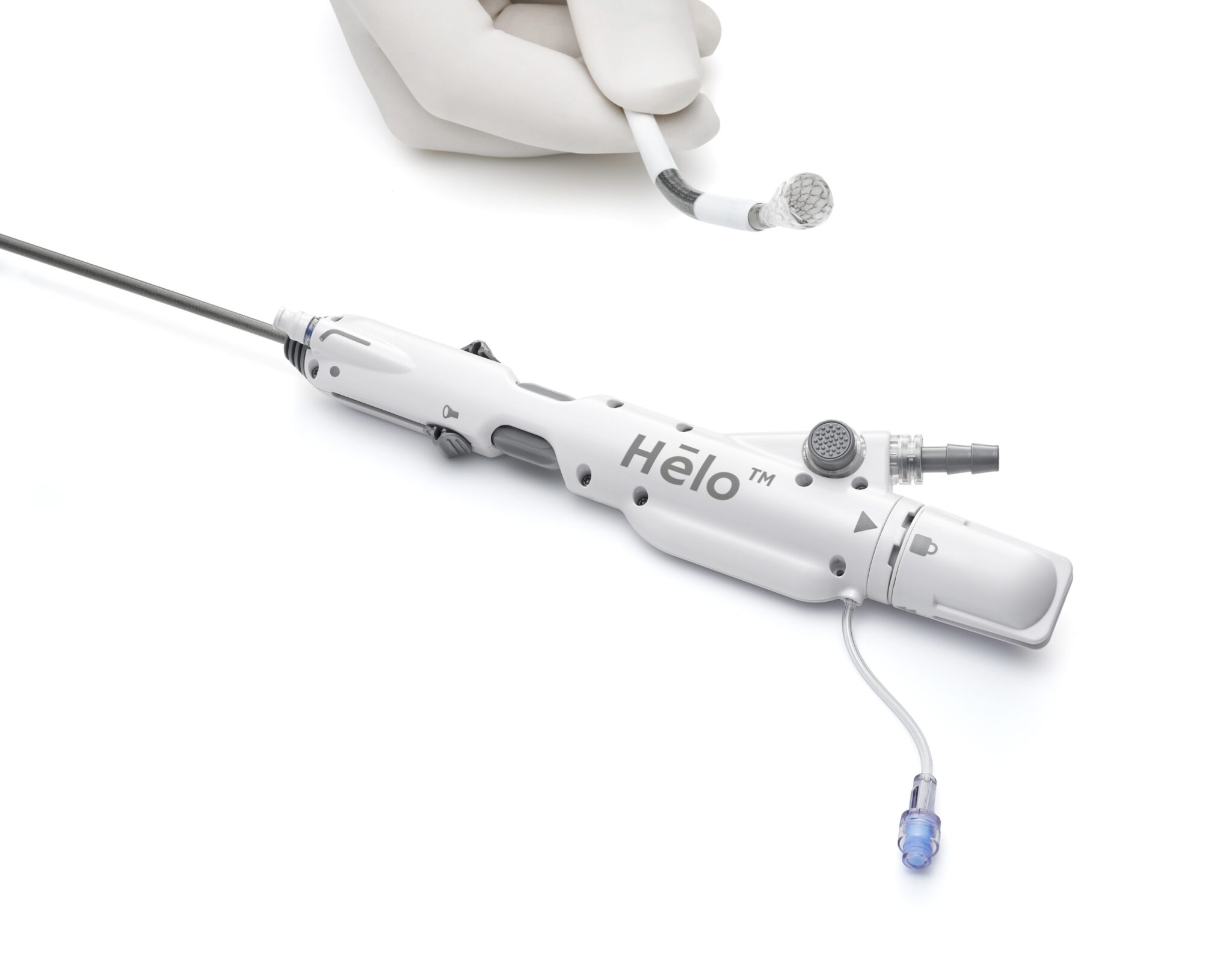
Endovascular Engineering’s (E2) next-generation mechanical thrombectomy (MT) platform is designed to eliminate trade-offs of existing systems. Its Hēlo system combines small-profile access with large-scale clot engagement, while integrating features like on-demand maceration and blood reinfusion to improve both efficacy and safety.
Today, we’re excited to share why Norwest co-led E2’s $80 million Series C round to support the company’s commercial launch and continued innovation. Norwest General Partner Dr. Zack Scott will also be joining E2’s board of directors.
Why Mechanical Thrombectomy is the New Standard of Care
Venous thromboembolism (VTE) is the umbrella condition that includes deep vein thrombosis (DVT), which occurs when a blood clot forms in the deep veins of the leg, and PE, which happens when part of that clot breaks loose and travels to the lungs.
Blood thinners, also known as anticoagulants, can slow clot progression but do not remove existing clots. For higher-risk patients, catheter-driven thrombolysis (CDT) can dissolve the clot, but the procedure carries significant bleeding risk and often requires a stay in the ICU.
Mechanical thrombectomy (MT) physically removes a clot through a catheter and has emerged as a transformative alternative. First‑generation MT systems, while groundbreaking, came with clear trade‑offs: they were either large‑bore devices that strain the heart or small‑bore devices that struggle to remove the clot, and nearly universal challenges with blood loss and procedural inefficiency.
When we asked more than 100 physicians across academic and community hospitals what their ideal MT system would look like, they described a platform that eliminates these compromises altogether — precisely the design philosophy that now defines E2. One doctor listed out the features they preferred, writing “Small bore, easy maneuverability, large aspiration capacity, efficacy, and safety with minimal blood loss.”
When we asked more than 100 physicians across academic and community hospitals what their ideal MT system would look like, they described a platform that eliminates these compromises altogether — precisely the design philosophy that now defines E2.
MT adoption has accelerated, driven by the formalization of care pathways through PE Response Teams (PERTs), expanding randomized controlled trial evidence, and growing recognition among hospitals of the clinical and economic value of rapid clot removal.
As MT evolves from a niche rescue therapy to a protocolized, hospital wide standard of care, this is the moment when second‑generation platforms, those that eliminate trade‑offs rather than force physicians to choose among them, can redefine the standard of care.
And this is precisely where E2 enters the story.
Eliminating Tradeoffs with Hēlo
E2 is a commercial‑stage medical device company developing a next‑generation MT platform designed specifically for VTE. The company’s flagship technology, the Hēlo® Thrombectomy System, demonstrates a deep understanding of the clinical realities and challenges physicians face every day.
E2 is a commercial‑stage medical device company developing a next‑generation MT platform designed specifically for VTE.
Its architecture is grounded in a powerful idea: go in small, but act big. The catheter enters the body through a small-bore access profile, which reduces instability of blood pressure and blood flow, and minimizes vascular trauma. Once inserted, it can then expand to a larger funnel capable of capturing large clots.
The system features a patented dual-action mechanism that integrates aspiration with mechanical clot disruption. And it integrates a blood return system that re-infuses aspirated blood in real time, preserving most of the aspirated blood, addressing blood loss, one of the most persistent frustrations with first‑generation devices.
Clinical Proof Meets Market Momentum
In the pivotal ENGULF study, Hēlo’s design translated into meaningful clinical performance. Right heart strain was significantly reduced, and physicians were able to remove clots efficiently, often on the first pass. Use of the blood return system was associated with minimal blood loss. Physicians repeatedly highlighted maneuverability and right-heart navigation as a standout advantage. The ability to move efficiently from right to left pulmonary arteries without repeated wire exchanges shortens procedure time and reduces instability in fragile patients.
In the pivotal ENGULF study, Hēlo’s design translated into meaningful clinical performance. Right heart strain was significantly reduced, and physicians were able to remove clots efficiently, often on the first pass.
E2 is entering the market at a pivotal moment. Adoption of MT for PE treatment presents a large and growing opportunity, with a TAM of $3+ billion with expected annual growth of 15- 20% over the next five years. Fueling this growth, hospitals are reevaluating their device portfolios as they prepare for guideline‑driven adoption of MT. At the same time, upcoming randomized trials — PEERLESS II and PE‑TRACT — are expected to expand MT use into broader intermediate‑risk populations, dramatically increasing the number of patients who may benefit from rapid clot removal.
The People and the Platform Poised to Redefine MT
Just as important as the technology is the team behind it. And in this case, the team starts with someone we’ve known and respected for years. We first engaged with CEO Dan Rose about five years ago, while he was leading his previous company, LimFlow. He stood out as a commercially minded operator who was deeply embedded with physicians and could articulate a clinical narrative with unusual clarity and conviction. We kept in touch and watched his success with E2. And when he began fundraising again, we were among the first calls he made.
Dan has assembled an exemplary leadership team with deep expertise across clinical research, regulatory strategy, catheter‑based engineering, salesforce development, and operational scale. Their experience is important as E2 builds its commercial organization, advances R&D, and scales operations.
We believe the company is uniquely positioned to become a leading MT platform for PE and an emerging force in DVT. The market is large, underpenetrated, and accelerating, and E2’s technology addresses key limitations of first‑generation systems.
Most important is the potentially profound impact E2’s technology can deliver for patients. Faster, safer, more predictable clot removal can mean fewer, or no, days in the ICU, fewer bleeding complications, and a better chance at recovery.
E2 is giving clinicians a tool that matches the urgency of the condition they are treating; a device that offers meaningful capabilities in a space long defined by compromise. We are proud to partner with E2 as they bring this platform to market and help shape the future of VTE care.